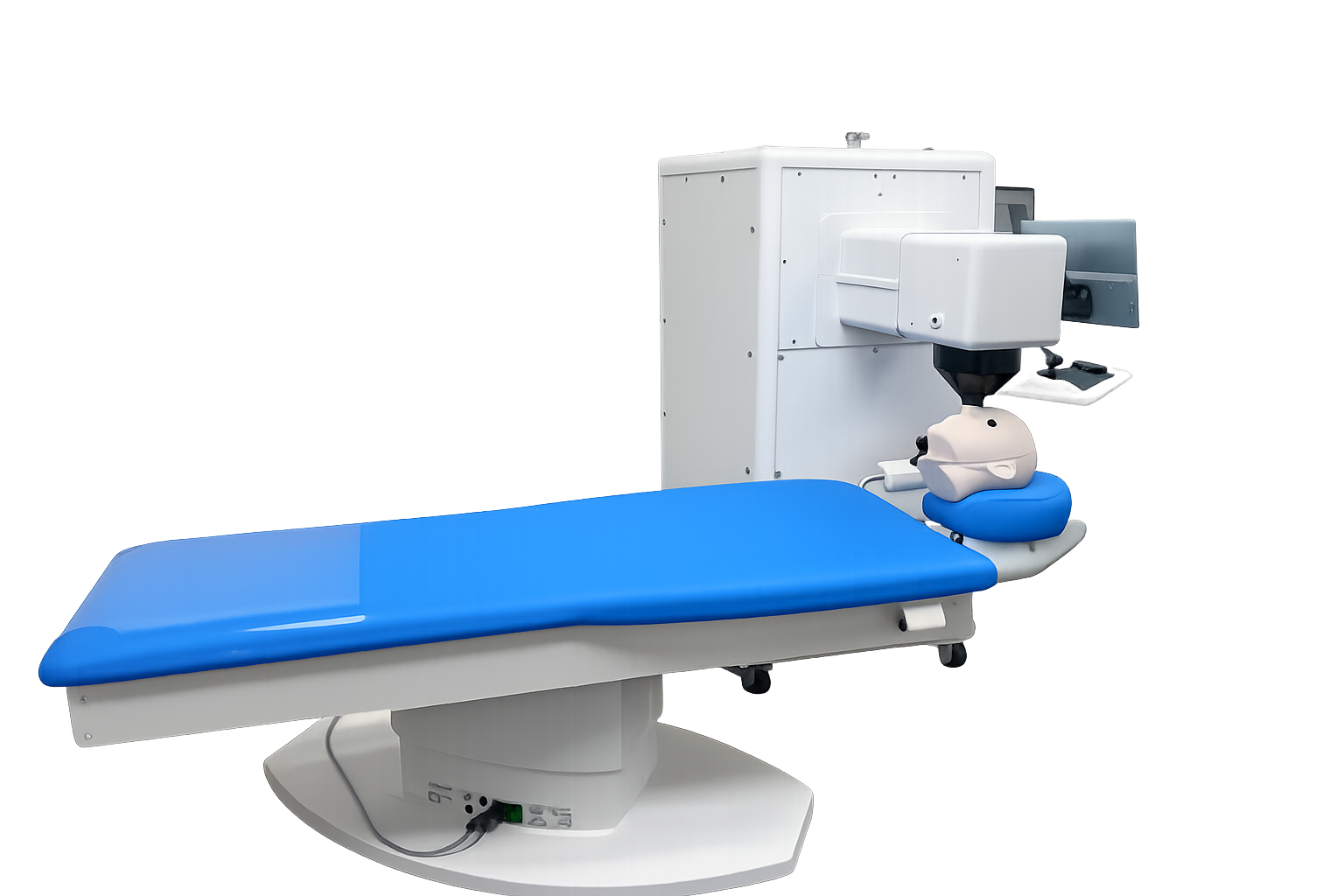
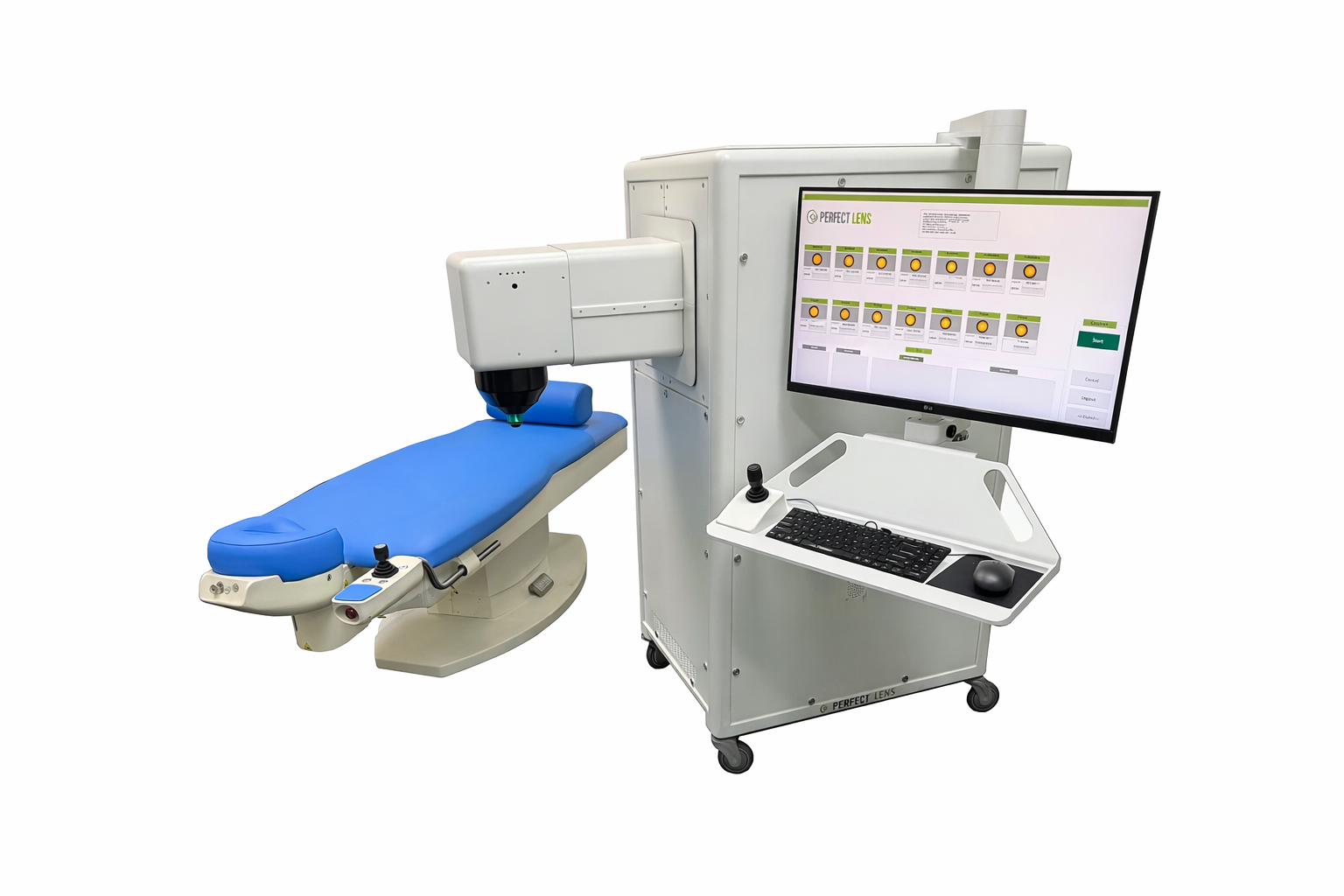
In vivo research addressing unprecedented precision requirements for post implant refractive adjustment
The in vivo research pathway represents a novel and investigational application of the RIS process,
enabling refractive modification after intraocular lens implantation. Unlike manufacturing systems,
no existing clinical platform provided the precision, stability, or optical control required for
this application.
Development of the in vivo system required rethinking every element of the workflow, including
patient docking, alignment, focus determination, treatment planning, and laser delivery.
Each subsystem was designed to meet precision requirements that exceed those of existing ophthalmic
laser platforms.
The research effort did not rely on adapting an existing clinical laser system, but instead required
the development of new concepts and validation strategies tailored to the unique optical and
mechanical constraints of treating an implanted lens inside the eye.
Ongoing research focuses on understanding sources of variability, defining safe operating windows,
and refining workflow robustness. The in vivo application is investigational and is being evaluated
through structured research and clinical study programs.
InVivoResearch
Investigational
PrecisionEngineering